7. Drug Update
Efruxifermin demonstrates potential in reversing liver scarring in MASH-reveals Phase 2b SYMMETRY trial
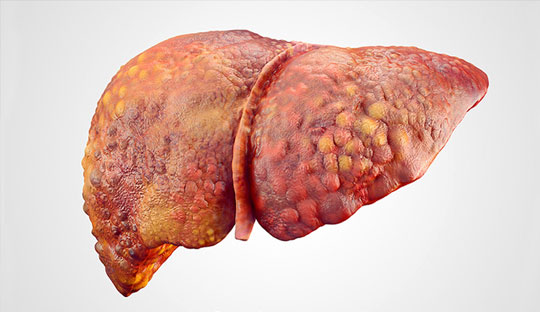
Akero Therapeutics recently reported promising findings from a placebo-controlled study showing its drug efruxifermin reversed liver scarring in patients with cirrhosis caused by the liver disease MASH. Efruxifermin is an experimental drug intended to replicate the biological action of fibroblast growth factor 21 (FGF21), which has the ability to control a variety of cellular functions and metabolic pathways.
The 96-week Phase 2b SYMMETRY study was a multicenter, randomized, double-blind, placebo-controlled, clinical trial that enrolled 182 patients with biopsy-confirmed compensated cirrhosis (F4, Child-Pugh class A) due to MASH who received once-weekly subcutaneous dosing of 28mg or 50mg EFX, or placebo. The primary efficacy endpoint for the study was the proportion of patients who achieve at least one-stage fibrosis improvement without worsening of MASH at week 36. Week 96 secondary measures included ≥1 stage fibrosis improvement and no worsening of MASH, MASH resolution, change from baseline in liver enzymes, noninvasive markers of liver fibrosis, glycemic control, and lipoproteins, as well as safety and tolerability measures.
Initial findings of the study demonstrated statistically significant reversal of fibrosis following 96 weeks of treatment with EFX. Among patients with baseline and week 96 biopsies, 39% of patients treated with 50mg EFX experienced reversal of cirrhosis with no worsening of MASH, compared with 15% for placebo. Further, the Intent to Treat (ITT) analysis with all missing week 96 biopsies treated as failures and without imputation revealed that 29% of the 50mg EFX group experienced reversal of cirrhosis with no worsening of MASH, compared with 12% for placebo. Histologically confirmed cirrhosis reversal was corroborated by improvements in key noninvasive measures of liver fibrosis and injury. Further, efruxifermin was shown to be generally well-tolerated.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




