The PaQ: An Insulin Pump for People with Type 2 Diabetes

The PaQ is an insulin pump designed for people with type 2 diabetes who might want to use insulin, but are deterred from the treatment option by the idea of taking multiple daily injections. Approved in Europe and awaiting regulatory approval in the United States, the small pump can be worn for up to three days. According to CeQur, the Switzerland-based company that makes the PaQ, the device holds up to 330 units of fast-acting insulin. This diabetes product has seven preset basal rates, push button bolus delivery, and can be worn in water to a depth up to six feet.
Novo Nordisk’s NovoPen Echo

Novo Nordisk is introducing the NovoPen Echo with a proprietary feature that not only records the time and dose amount when delivered, but also allows injections in half-unit increments. The pen is specially designed to appeal to children, and not only because it keep s might a record of last doses that busy children not recall. The pen is designed to be cool looking. It comes in red and blue but also has many “skins,” or graphically intense overlays available, giving kids a ton of changeable options to customize their pens to their personal tastes and preferences.
The NovoPen Echo was approved by the FDA in August 2013 and is expected to hit the U.S. market in early 2014. The NovoPen Echo is already available in Europe, Canada, and Israel.
Affrezza: An Ultra-Rapid-Acting Inhaled Insulin
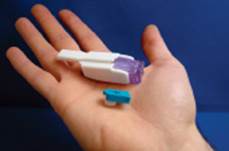
This diabetes product could be a real game changer if approved by the FDA. MannKind Corporation’s inhalable insulin called Affreza may be reaching the U.S. market in 2014.
The system delivers what the company calls an “ultra rapid-acting mealtime insulin therapy” to be taken in conjunction with food by either type 1 or type 2 diabetics. The inhalable powder is delivered in premeasured, single use cartridges by using a proprietary inhaler that fits comfortably into the palm of your hand.
MannKind resubmitted it’s New Drug Application to the FDA in October and the FDA is expected to finish its review of Affreza by April 2014.
The Dario Glucose Meter

The Dario Glucose MeterLabStyle Innovations’ Dario is an all-in-one system that can make managing your diabetes easier and more convenient. This was one of the diabetes products that really caught the eye last year. It’s not only a blood glucose meter with a built-in lancing device that includes a compartment storing up to 25 test strips, but it also interacts with your smartphone.
A breakaway section of the device connects to your smartphone via the headphone connection and you’re connected to a Dario app. Once connected, you just add blood to the test strip and, presto! The results then appear on your phone. From there at the touch of a button you can share the results with friends, family, medical professionals, and others. This product was expected to be available in 2013, but it is now expected to be available in early 2014 in Europe, and LabStyle Innovations predicts the device will make its debut in the United States later in the year.
ShugaTrak: An App For Sharing Blood Glucose Results

ShugaTrak is a new mobile app for people with diabetes that, at the start of 2014, is already available. When someone with diabetes checks blood glucose, ShugaTrak gets the reading from the meter using a Bluetooth adaptor, stores it in an online database, and transmits it to interested parties like family and friends in text messages and emails. This all happens immediately, and the person with diabetes doesn’t have to do anything beyond an ordinary blood glucose check. Shugatrak gives peace of mind to parents, spouses, and adult children of people with diabetes by informing them of their loved ones’ blood glucose readings when they are apart.
Read More





