3. Can a Diabetes Drug Help Prevent Opioid Overdoses? Semaglutide Shows Promise
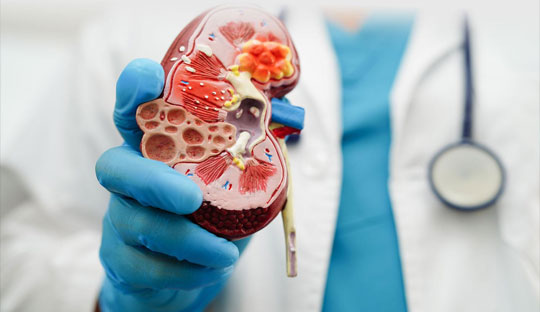
The EMPA-KIDNEY trial had revealed that empagliflozin, a sodium–glucose cotransporter 2 (SGLT2) inhibitor, had positive cardiorenal effects in patients with chronic kidney disease who were at risk for disease progression. Recently the research team had conducted post-trial follow-up to assess the effects of empagliflozin after the discontinuation of the trial drug.
In the active trial, patients with chronic kidney disease were randomly assigned to receive either empagliflozin (10 mg once daily) or matching placebo and were followed for a median of 2 years. Surviving patients who consented were observed for 2 additional years and they were not administered either trial empagliflozin or placebo during the post-trial period. However, the local practitioners could prescribe open-label SGLT2 inhibitors, including open-label empagliflozin. The primary composite outcome was kidney disease progression or cardiovascular death as assessed from the beginning of the active-trial period to the end of the post-trial period.
4891 people were enrolled in the post-trial period. During the combined active- and post-trial periods, a primary-outcome event occurred in 865 of 3304 patients in the empagliflozin group and in 1001 of 3305 patients in the placebo group. During the post-trial period only, the hazard ratio for a primary-outcome event was 0.87. During the combined periods, the risk of kidney disease progression was 23.5% in the empagliflozin group and 27.1% in the placebo group; the risk of the composite of death or end-stage kidney disease was 16.9% and 19.6%, respectively; and the risk of cardiovascular death was 3.8% and 4.9%, respectively. There was no effect of empagliflozin on death from noncardiovascular causes in both groups.
The study published in ‘The New England Journal of Medicine’ concluded that for up to a year following its discontinuation, empagliflozin maintained additional cardiorenal benefits in a wide range of individuals with chronic kidney disease at risk for advancement.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




