7. Drug Updates
Finerenone in Type 1 Diabetes with CKD
Finerenone, a selective nonsteroidal mineralocorticoid receptor antagonist, has previously demonstrated significant renal and cardiovascular benefits in individuals with type 2 diabetes and chronic kidney disease (CKD). Emerging evidence from a recent phase 3 clinical trial now explores its role in people with type 1 diabetes and CKD, a group with limited targeted therapeutic options beyond standard renin–angiotensin system blockade.
In this randomized trial involving adults with type 1 diabetes, reduced eGFR, and significant albuminuria, finerenone therapy led to a clinically meaningful reduction in urinary albumin-to-creatinine ratio (UACR), a key marker of kidney damage. Over a 6-month period, UACR decreased by 34% in the finerenone group compared to 12% with placebo, translating to a 25% greater reduction. These findings indicate a meaningful improvement in renal risk markers, suggesting potential for slowing disease progression.
A modest initial decline in eGFR was observed with finerenone, a pattern consistent with hemodynamic effects seen in other reno-protective agents, with values trending back toward baseline after treatment discontinuation. The most commonly reported adverse event was hyperkalemia, occurring in about 10% of participants, though treatment discontinuation due to this was infrequent. Overall, finerenone was generally well tolerated. These results highlight finerenone as a promising addition to the therapeutic armamentarium for CKD management in type 1 diabetes. However, longer-term studies are needed to confirm sustained renal benefits, cardiovascular outcomes, and long-term safety before routine clinical adoption.
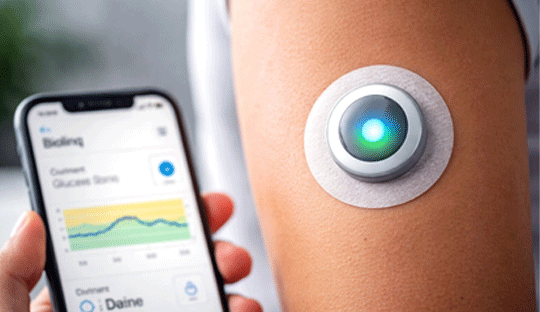
Biolinq Shine – A Needle-Free Approach to Glucose Monitoring
The Biolinq Shine introduces an innovative, needle-free approach to glucose monitoring, offering a more comfortable alternative for adults with type 2 diabetes who are not using insulin. Recently cleared by the U.S. FDA, this compact, quarter-sized wearable patch is designed to address one of the major limitations of conventional continuous glucose monitoring (CGM) systems, the need for subcutaneous sensor insertion, which can be a deterrent for many users.
Unlike traditional CGMs that provide continuous numerical glucose readings, Biolinq Shine simplifies monitoring through a color-coded LED indicator that visually signals whether glucose levels are within, above, or below the target range. This intuitive design reduces data overload and may enhance user engagement, particularly for individuals who do not require intensive glucose tracking. The device is worn on the skin like an adhesive patch and integrates with a mobile application, where users can access more detailed insights, including trends related to glucose patterns, physical activity, and sleep.
While the system does not offer the same level of precision or detailed analytics as standard CGMs, it addresses an important gap in diabetes care by providing a minimally invasive, easy-to-use option for glucose awareness. This makes it particularly suitable for individuals with type 2 diabetes managed without insulin, where the focus is often on general glucose trends rather than minute-to-minute fluctuations. Overall, Biolinq Shine represents a meaningful step toward more patient-friendly, accessible glucose monitoring solutions, with the potential to improve adherence and broaden the adoption of digital health tools in diabetes management.
For enquiries info@jothydev.net.
Please visit: jothydev.net | research.jothydev.com | diabscreenkerala.net | jothydev.com/newsletter




